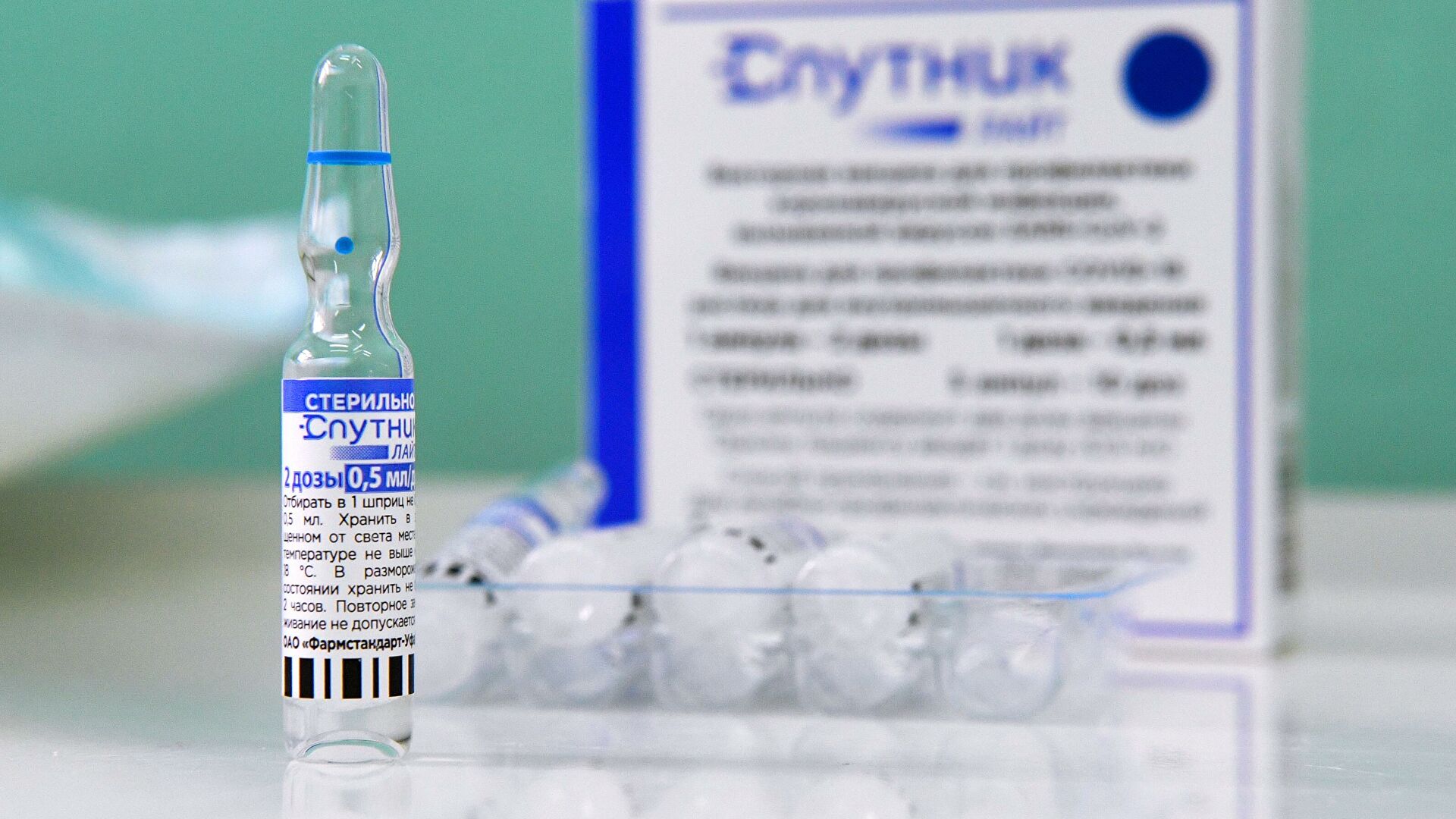
Sputnik Light vaccine ampoule MOSCOW, February 6 Sputnik Light single-component vaccine registered in India, thus, more than 2.5 billion people now live in countries that have approved Sputnik Light as a stand-alone vaccine and as a universal booster, RDIF said. «The Russian Direct Investment Fund announces registration by the Drug Controller General of India , DCGI) of the single-component Sputnik Light coronavirus vaccine. Thus, Sputnik Light has been approved in more than 30 countries with a total population of over 2.5 billion people,» the foundation said. Vechorko reported an increase in the number of patients with the omicron strain. As a universal booster, the drug has already been approved in a number of countries, including Argentina, Bahrain, the United Arab Emirates, San Marino, and the Philippines, the fund recalled. The Sputnik Light vaccine as a booster significantly increases the virus-neutralizing activity against the Omicron strain to levels comparable to the titers that Sputnik V forms against the original strain. The Sputnik Light vaccine is the first component (recombinant human adenovirus serotype 26 (rAd26 ) Sputnik V. The single-component regimen of Sputnik Light has a number of key advantages, including a simple vaccination process, as well as the ability to increase the effectiveness and duration of action of other vaccines when used as a booster injection.
Vechorko reported an increase in the number of patients with the omicron strain. As a universal booster, the drug has already been approved in a number of countries, including Argentina, Bahrain, the United Arab Emirates, San Marino, and the Philippines, the fund recalled. The Sputnik Light vaccine as a booster significantly increases the virus-neutralizing activity against the Omicron strain to levels comparable to the titers that Sputnik V forms against the original strain. The Sputnik Light vaccine is the first component (recombinant human adenovirus serotype 26 (rAd26 ) Sputnik V. The single-component regimen of Sputnik Light has a number of key advantages, including a simple vaccination process, as well as the ability to increase the effectiveness and duration of action of other vaccines when used as a booster injection. Named six skin symptoms of «omicron» that may not go away for a long time Reddy's Laboratories, conducted additional clinical trials of Sputnik Light in India, the positive data of which were also taken into account by DCGI when registering. Clinical studies and data on vaccination of the population in many countries have demonstrated the high safety and effectiveness of Sputnik Light when used as a stand-alone vaccine, and a universal booster. The approval of Sputnik Light will expand India's national vaccine portfolio, which included Sputnik V in April 2021. India is the leading manufacturing hub for Sputnik V. Sputnik V and Sputnik Light are based on a well-established human adenoviral vector platform and are not associated with serious post-vaccination adverse events such as myocarditis or pericarditis. The high safety and effectiveness of Sputnik Light and Sputnik V has been confirmed by more than 30 studies and vaccination data from more than 60 countries. :22InfographicComparison of effectiveness of Sputnik V and Pfizer vaccines against omicron-strainInfographicView
Named six skin symptoms of «omicron» that may not go away for a long time Reddy's Laboratories, conducted additional clinical trials of Sputnik Light in India, the positive data of which were also taken into account by DCGI when registering. Clinical studies and data on vaccination of the population in many countries have demonstrated the high safety and effectiveness of Sputnik Light when used as a stand-alone vaccine, and a universal booster. The approval of Sputnik Light will expand India's national vaccine portfolio, which included Sputnik V in April 2021. India is the leading manufacturing hub for Sputnik V. Sputnik V and Sputnik Light are based on a well-established human adenoviral vector platform and are not associated with serious post-vaccination adverse events such as myocarditis or pericarditis. The high safety and effectiveness of Sputnik Light and Sputnik V has been confirmed by more than 30 studies and vaccination data from more than 60 countries. :22InfographicComparison of effectiveness of Sputnik V and Pfizer vaccines against omicron-strainInfographicView

